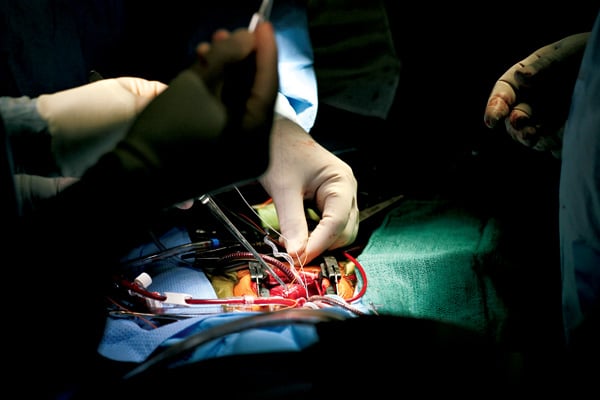
The fetal monitor throughout the night hasn’t indicated that her heart is in distress, nor did a routine ultrasound exam several weeks earlier detect any cardiac abnormality. But the moment Taylor Keaveney is born on a rainy night in April 2010, the dark-blue cast of her skin and her ominously low blood-oxygen level reveal that her life is in peril.
Doctors and nurses at Reston Hospital Center rush the infant from the delivery suite to the neonatal intensive-care unit (NICU) so quickly that her mother, Amanda, exhausted from a long labor, barely catches a glimpse of her baby. Her husband, Joe, is shaken by the sight of his daughter but doesn’t betray his worry to his wife.
Dr. Sharon Karr is at a Virginia pediatric-society meeting in Tysons Corner when she answers a page and then calls the neonatologist at the hospital.
“We have a blue baby here,” the neonatologist tells Karr, a pediatric cardiologist. “We took x-rays, and it looks like she has a total anomalous pulmonary venous return.”
Most babies born with congenital heart defects—occurring in about 1 in 125 live births—are not in immediate danger. But of all the congenital heart defects, three can be true emergencies: a type of hypoplastic left heart, in which there’s only one pumping chamber instead of two; a tight aortic narrowing that can’t be opened with medication; and baby Taylor’s defect, a total anomalous pulmonary venous return.
I take a special interest in Taylor’s case and in congenital heart defects generally because I was born with one. At my request, the administration and medical staff at Inova Fairfax Hospital for Children have allowed me to observe all phases of its heart program. Over several months I witness heart surgeries, activities in the catheterization lab, and mortality-and-morbidity conferences. In those months, I observe the newest technologies for repairing young hearts—and reflect back on my own heart and how it was repaired 50 years ago this year. Medicine has come a long way.
After getting the diagnosis from the neonatologist, Karr quickly leaves her meeting, drives 20 minutes to Reston Hospital Center, and hurries to the NICU. The neonatologists have put Taylor on a ventilator and turned up the oxygen to 100 percent, but she remains dark blue with a blood-oxygen level of 50 percent when it should be in the 90s. Karr puts her stethoscope on the infant’s chest. The first thump of the heartbeat sounds normal, but the second thump is unusually loud.
She slides a wand-like transducer over Taylor’s frail chest to get an echocardiogram image of the heart and to study the flow of blood through the major vessels. Taylor’s left heart appears small and compressed, almost flattened; her right heart is dilated and not functioning normally. The echocardiogram confirms the diagnosis: The pulmonary veins from Taylor’s lungs to her heart never connected during fetal development.
This defect arose when Taylor’s heart began to form during the fifth week of her fetal life. This is the most critical phase of embryonic heart development, when a primitive tube of myocardial cells folds to create the heart’s four chambers.
No bigger than the head of a small nail at first, the heart continues to develop, and by ten weeks’ gestation its four chambers can be visualized by ultrasound. But a genetic glitch can malform the heart’s complex architecture—resulting in a valve that doesn’t open properly, vessels attached to the wrong chambers, a hole in the heart wall, or a condition such as Taylor’s.
“When you consider just how complex the heart is,” Karr says, “it’s almost a miracle that so many develop normally.”
It’s not surprising that Taylor’s heart defect was missed on a routine ultrasound months before her birth, Karr says. In such examinations, technicians usually confirm that the heart has four chambers and normal outflow vessels, but because the pulmonary veins are very hard to image, only a pediatric cardiologist well trained in fetal ultrasound is likely to have detected Taylor’s defect in utero.
Studying the images of Taylor’s miniature heart, Karr sees that the four pulmonary veins—two from the left lung and two from the right—come together in a confluence unconnected to her heart. Moreover, Taylor’s spaghetti-size pulmonary veins are tiny even for an infant, making any surgical repair that much more delicate. Karr knows Taylor will die, and soon, without surgery.
Around 9 pm, Karr calls Dr. Irving Shen, director of pediatric cardiac surgery at Inova, who is on call that night. In his four years at Inova, Shen has earned the trust of Karr and other cardiologists. She reaches him as he’s about to get ready for bed and tells him Taylor’s condition.
“She needs to go to the OR tonight,” Karr says.
Shen asks Karr to bring Taylor to Fairfax Inova and gets on the phone to assemble the cardiac-surgery team. Most are at home after having already put in a long day in the operating room.
In Amanda’s hospital room, Karr tells Amanda and Joe that their daughter is gravely ill. “She has to be operated on right away,” Karr says, “or we could lose her.”

Amanda is stunned. She thought Taylor had a lung problem that could be corrected. The news that her baby suffers from a critical heart problem is almost overwhelming.
“I’ve already phoned Dr. Shen, and he is on call and available,” Karr tells the couple. “If you’re going to send her anywhere, send her to Inova. He’s the best.”
Amanda, like Joe a practicing Catholic, has one request: “I want Taylor to be baptized before you transport her to Inova.”
A hospital priest arrives and Amanda, still numb from the waist down, is wheeled to the NICU, where she sees her newborn daughter for the first time. As the priest performs the baptism, Joe puts his arm around Amanda; she slides her hand inside Taylor’s isolette to hold the baby’s tiny hand, stifling tears and wondering if she’ll see her baby alive again.
Amanda wants to go to Inova Fairfax. “If my daughter is going to die,” she says, “I want to be there with her.” But the doctors tell her she has to stay at Reston until she herself recovers from the delivery.
Amanda returns to her hospital bed. Joe’s sister Kathleen has arrived, and she drives him to Fairfax Inova.
Joe and Amanda, both in their mid-thirties, already have been through a series of hard experiences. Joe, a construction supervisor, was laid off during the real-estate slowdown of 2008. A few months later, Amanda lost her job in a medical billing office. The couple exhausted their savings, declared bankruptcy, and eventually lost their house in Stephens City, Virginia. They moved in with Amanda’s parents in Sterling and transferred their two older daughters, Abigail and Madison, to a new school.
Joe, who was born in Silver Spring, can’t shake the feeling that he has failed his family. Now, riding in his sister’s car along Route 267 through the drizzly blur of oncoming headlights, he says he must have done something wrong for all this to befall his family.
“This didn’t happen to you because you did something wrong,” Kathleen says. “This is life—this is what happens.”
Kathleen suggests that they pray as they drive toward Inova Fairfax Hospital.
Irving Shen slips into his rain gear, says goodbye to his wife, and walks downstairs to his garage to retrieve his bicycle. Rain or shine, night or day, Shen either rides his bike or walks the three miles from his townhouse to Inova Fairfax Hospital. He usually leaves at 5:30 am when he walks, 5:45 when he rides.
“When I walk or ride my bike to the hospital in the morning,” he says, “I think about the cases I have that day. When I return at night, it gives me time to reflect on what’s happened so I can unwind before I get home.”
As he pedals along the familiar trail in the rainy darkness, he thinks about the emergency he faces with little Taylor. Shen did much of his surgical training in Oregon and Washington state, where, for reasons that aren’t clear, he saw a higher-than-average incidence of the congenital heart defect that Taylor has. This gave Shen more experience in the procedure than he might have otherwise acquired.
A slender man of medium height with delicate hands and fingers that could belong to a violinist, Shen sports a ponytail and wears sneakers. He laughs easily, often at himself. His office is as unpretentious as he is, complete with couch and blankets.
Shen came to the United States from Hong Kong in 1973 as an 11-year-old during China’s cultural revolution, the social and political upheaval that spilled over into Hong Kong. Shen’s parents, both from mainland China, feared for their children’s future, so they brought Irving and his three sisters to this country, settling in Oregon. Shen’s father, an accountant in Hong Kong, found work in Oregon keeping the books for a charitable organization. He had named his son Irving because the Irving Trust bank stood next to the hospital where the boy was born and he liked the name.
Irving Shen began learning English and entered the school system, soon establishing himself as an academic star. He graduated from Cornell, then returned to Oregon for medical school. After his general-surgery residency, he wanted to learn how medicine was practiced in other countries, so in 1995 he and his wife, Carmen—a nurse he’d met during training at the University of Washington Hospital in Seattle—moved to New Zealand. Still uncertain of what he wanted to specialize in, Shen operated in a pediatric cardiac-surgical service under a chief notorious for terrifying medical students and residents.
One day, Shen scrubbed in with the chief to operate on a young child with tetralogy of Fallot, a complex congenital heart defect. A medical student scrubbed in, too, and as usual the attending physician peppered the student with questions about the defect. Shen, amazed that the student correctly answered every question thrown at him, learned that the med student had been born with tetralogy of Fallot and had been operated on as a young boy.
“That was a turning point for me,” Shen says, “because I could see how this student’s entire life had been impacted profoundly by the repair of his congenital defect. From that point on, I knew I wanted to become a pediatric heart surgeon.”
When Inova Fairfax began looking for a new pediatric heart surgeon, Shen’s name and reputation came to the fore, and the hospital recruited him from Oregon Health & Science University, where he was an associate professor of surgery. It soon became apparent to his new colleagues at Inova that the soft-spoken Shen didn’t shy away from an argument or a difficult case.
>> Next: Worrying About Surprises
Worrying About Surprises
As he rides his bike, careful to avoid slipping on the wet pavement, Shen visualizes the baby’s heart in three dimensions and keeps replaying the impending surgery in his mind, recalling previous ones he has performed, trying to anticipate any surprises.
Asked whether he thinks the surgeries through in English or his native Chinese, Shen answers: “Neither. I don’t think in language; I think in images, like a gymnast who rehearses his routine in his mind before he performs.”
This visualization process allows Shen to remain calm under the most trying circumstances in the operating room. “Some surgeons may be able to wing it if something goes wrong,” he says, “but I need to have the entire procedure plotted out in my mind, including unanticipated complications that might happen, so when something out of the ordinary occurs, I will know what to do.”
This is the only way, says Shen—who invests so much emotion in his young patients that he and his wife decided not to have children of their own—that he can face himself if something goes wrong.
By the time Shen arrives at the hospital, he has mentally rehearsed the surgery he’ll perform on Taylor. He walks to the waiting area, introduces himself to Joe and his sister, and, using a softball-size model of a human heart, explains what’s wrong with Taylor.
The heart has two sides with two basic functions. The “right heart” is composed of the sac-like atrium that collects blood depleted of oxygen after its circuit around the body as well as the muscular ventricle that pumps this bluish-colored blood to the lungs, where carbon dioxide is exchanged for the oxygen we inhale when we breathe. The lungs then send this red, freshly oxygenated blood to the left atrium, which in turn sends it to the left ventricle, the heart’s most powerful pumping chamber. From there, it’s propelled through the aorta into the arteries and then to every cell in the body. In a healthy adult, the left ventricle can pump six liters of blood a minute.
In Taylor’s case, the pulmonary veins that return oxygenated blood from the lungs back to her “left heart” aren’t connected to her heart, so her left heart receives virtually no oxygenated blood from her lungs.
Two things prevented Taylor from being stillborn and continue to keep her alive, at least for the moment: One is a hole in her septum, the wall that separates the left and right atria. The hole allows blood to flow into her left heart, where her left ventricle pumps some partially oxygenated blood out to her body’s organs.
The second thing is a vertical vein that carries blood directly from the lungs to the right heart during fetal development. This vein closes when the pulmonary veins connect to the heart. But because Taylor’s pulmonary veins hadn’t connected, her vertical vein never shut down, and it now provides critically needed oxygenated blood. But it’s so badly obstructed that it carries only a trickle of oxygenated blood to her heart.
The precise name for Taylor’s defect is “obstructed total anomalous pulmonary venous return.” It also means that Taylor, all 5 pounds and 11 ounces of her, teeters on the brink of death from her heart defect and from the pressure exerted back on her lungs, pressure so high that it doesn’t allow the blood to absorb oxygen normally.
Every cell in the body needs oxygen to survive, but none as much as heart cells. Starved of sufficient oxygen, the brain can remain unharmed for minutes, but the relentlessly beating heart cannot. Its myocardial cells need ten times more oxygen than other organs of the body. If its oxygen needs aren’t met, it beats even faster in an attempt to get it, an action that requires yet more oxygen. Caught in this cycle, Taylor’s blood will become more acidotic from oxygen deprivation and her heart’s myocardial cells will begin to die. Before long, her heart will arrest or be damaged beyond repair.
To guard against the worst, Karr remains with the infant in the Inova pediatric ICU.
After Shen has explained all that’s wrong with Taylor, he reminds Joe that the surgery entails significant risk. He then lays the heart model on the floor.
“Now I’m going to tell you something, and please don’t get upset,” Shen says, “but your daughter’s heart is the size of a walnut. That’s the space we have to work in.”
By the time Shen leaves, Joe is convinced that his daughter’s life is in the best possible hands.
Born at the Right Time
Anesthesiology has been described as hours of boredom intermingled with moments of terror. As Dr. Erin Foley drives around the Beltway toward the hospital, knowing that this will be an especially challenging case, she reminds herself of her training at Children’s Hospital Boston. It has prepared her for any moments of terror that might lie ahead. I have to be on my A-game tonight, she tells herself.
In the OR, Foley works at the head of the bed amid a cluster of state-of-the-art equipment to monitor Taylor’s physiological functions—from blood pressure, body temperature, and heart rate to the pressure in her left atrium. If something goes wrong during surgery, Foley probably will be the first to know.
Bathed in a white light, the operating room has the sharp aroma of the antiseptic betadine. The room’s temperature hovers near 60 to help reduce the patient’s body temperature and metabolic rate, the ever-present threat in any surgical procedure, as Taylor is wheeled in. She’s still on the ventilator she has been on since being intubated at Reston Hospital Center, but almost immediately Foley removes her endotracheal tube and replaces it with a larger one to allow better suction of secretions and blood during surgery.
She places a monitoring pad on Taylor’s forehead and a second pad on her flank. These will allow her to monitor oxygen saturation in different regions of Taylor’s brain as well as her kidneys through an INVOS monitor that uses infrared technology.
Humming in the background is the muffled sound of soft-rock music and the low murmur of people making preparations for surgery. Taylor was born at the right time. As recently as the early 1980s, many babies who were as sick as she was were given compassionate care and little else. Some were too small to be operated on or had hearts that doctors knew wouldn’t function for long. They went home and died.
That changed in the late 1980s and early ’90s as refinements in the heart-lung machine, new microsurgical instrumentation, and improved imaging meant that nearly all children with congenital heart defects could be offered surgery. Even babies weighing less than two pounds are now placed on heart-lung bypass, something almost unheard of 15 years ago.
Gleaming instruments are arrayed on sterile blue towels in the order Shen has requested. He stands on Taylor’s right side, the “headlight” attached to his forehead aiming a brilliant beam of light wherever his eyes scan. A small camera attached to the headset displays the operation on overhead screens.
Heart surgery is a form of violence on the human body, an incursion into skin, bone, and tissue to expose and repair the small part of it that failed to develop the way it shou
ld. Be it a nonfunctioning valve, the narrowing of an artery, or any of dozens of possible defects, it needs to be repaired or else the child is consigned to an abbreviated life, if any life at all.
The second-century Greek physician Galen was the first to report seeing a living human heart within an open chest when he removed part of the sternum from a wounded Roman servant. Understanding how the heart functioned had to wait until English physician William Harvey published On the Motion of the Heart and Blood in Animals in 1628. Harvey’s description of the heart as a powerful, four-chambered pump that continually circulates blood throughout the body by means of a multitude of vessels became the foundation for modern cardiovascular research.
Because the heart is critical to life, physicians and scientists well into the 20th century believed it could never undergo surgery. Surgeons began to operate on the heart, the last organ in the human body to yield to surgery, in the 1940s and ’50s.
Like Taylor, I was born at a propitious time. In 1961, when I was 22 and my time was running out, technology had advanced to the point where open-heart surgery was feasible, and the chance of success in cases like mine were greater than the risk of failure.
Taylor lies still, her eyes taped shut, as a nurse swabs her chest with antiseptic. She remains in critical but stable condition. Foley has given her isoflurane and fentanyl for general anesthesia; during the course of the operation, she’ll administer more than a dozen drugs to maintain the infant in optimal condition. She also monitors Taylor’s blood loss and blood replacement.
After midnight, with the cardiac team of about a dozen people in place, Shen makes a four-inch-long vertical incision in the middle of Taylor’s chest, starting it as low as possible so Taylor won’t have a visible scar when she wears scoop-neck dresses later on. He cuts with a pen-size cautery that coagulates tiny blood vessels with a gentle electrical current, emitting tiny wisps of smoke and an acrid smell.
Following the incision line, Shen guides a surgical saw that makes a muted buzzing sound as it cuts through Taylor’s exposed sternum. With well-choreographed steps, he inserts a retractor to hold the chest incision open, then retracts the thymus gland to gain the exposure needed to hook her up to the heart-lung bypass machine.
Shen works with a surgeon’s detached concern, keeping his emotions at a distance while concentrating on—and caring deeply about—his patient. There’s often light banter in the OR, but it’s late and the surgery too perilous for small talk on this night.
>> Next: “A Phenomenal Surgeon”
“A Phenomenal Surgeon”
In past years, the pediatric heart program at Inova Fairfax had been considered solid, but it rose to a new level with Shen’s 2006 arrival. Karr calls him “a phenomenal surgeon.”
Trained to operate on the most complex congenital heart defects, Shen has broadened the scope and depth of the Inova pediatric cardiac program. He formed a partnership with fellow cardiac surgeon Lucas Collazo, who has been at Inova since 1999 and is in his mid-forties.
“Irving has just the right balance of ego, security, and humility and is a delight to work with, and so is Lucas Collazo,” says pediatric cardiologist Thomas Hougen, a professor of pediatrics at Georgetown University who refers many heart patients to Inova Fairfax. “They make a very good team. The medical students I send over there are always surprised that two top-flight surgeons are so easy to get along with. Their experience at other places is not like that.”
Dr. Hougen says training alone doesn’t create a great surgeon. “It’s a combination of things. Some surgeons have it—and I’ve seen others train for years and never attain it.”
The mutual support between Drs. Shen and Collazo is important because the attitudes of the surgeons determine how well the support staff—nurses, technicians, and the perfusionists who operate the heart-lung bypass machine—perform their jobs. In virtually every conversation I have with them, the two surgeons credit the entire cardiac team for any success they’ve had.
Although Inova Fairfax isn’t as well known as the country’s premier pediatric heart programs at Children’s Hospital of Philadelphia and Children’s Hospital Boston, results for the more than 300 young patients who annually undergo heart surgery in Inova’s pediatric-surgery program are published every year and rival those anywhere.
As Shen navigates the miniature landscape of Taylor’s chest cavity, he’s aided by physician assistant Tom Lauer. Shen exposes Taylor’s plum-colored, pulsating heart. With surgical scissors, he delicately cuts open her pericardium, the thin, translucent sac encasing her heart. She has been given heparin, an anticoagulant, to prevent clotting. He then inserts plastic cannulas into her vena cava and aorta, the body’s two largest vessels, which connect Taylor to the heart-lung bypass machine. He loops sutures to tighten the seal and hold the cannulas in place.
The perfusionist circulates Taylor’s blood through a cooling coil in the heart-lung machine, a process that drops her body temperature to 27 degrees Celsius (about 80 Fahrenheit) to slow her metabolism and reduce the demand for oxygen in her tissues, especially during the time Taylor’s heart is stopped and the heart-lung machine—“the pump”—circulates her blood. Unlike the heart, which pulses blood when it beats, the pump streams blood continuously.
Shen clamps the aorta to stop the blood flow and signals the perfusionist to inject potassium into an IV line inserted directly into Taylor’s heart. The tiny organ, throbbing only moments earlier, is stilled in less than half a minute as the potassium shuts down its electric circuitry. The EKG monitor flatlines, and her abruptly stilled heart becomes flaccid as the heart-lung bypass machine takes over, eliminating the major fear—that her heart will suddenly fail.
Shen cuts into the fibrous heart muscle itself and suctions blood from inside the heart chambers to gain better access to the atrium. Karr watches on an overhead monitor as Shen incises a precise opening in the back of the atrium before undertaking the meticulous task of maneuvering into place the four unconnected pulmonary veins, all conjoined into one, and suturing them to the atrium with a series of intricate stitches. Throughout the procedure, Foley periodically reports that Taylor’s vital signs are stable.
At about 5 am, with the connection complete and Taylor’s heart sewn back together, the room’s temperature is raised to 65 and Taylor’s blood is warmed by the heart-lung machine’s heat exchanger. As the surgery team watches, Taylor’s heart stirs to life as they wean her off bypass. For the 164 minutes that Taylor was on bypass, her heart was effectively dead. Its beating seems almost like a resurrection.
Karr performs a post-op echocardiogram while Taylor is on the operating table. The infant’s right heart, once dilated and abnormal, now appears nearly normal. Her left ventricle is small, unused to the copious amount of blood it now has to pump. It will take a few days for it to adjust. Karr is elated to observe normal blood flow through the pulmonary veins and into the left atrium—a perfect connection.
Relieved of the stress of monitoring Taylor through more than four hours of surgery, Foley is thrilled by the surgery’s success.
Because swelling can occur postoperatively, Shen doesn’t close Taylor’s chest immediately, leaving the heart room to expand.
Before sunrise, he meets with Joe in the pediatric ICU, where Taylor lies sleeping in a small bed, still on the ventilator amid a nest of tubes and lines. Bandages cover her open chest while behind her head a wall of monitors flickers and beeps.
Shen tells Joe that the surgery went well, Taylor’s heart is repaired, and her condition is critical but stable.
“Will she need any more surgery?” Joe asks.
“I feel confident she will not need any more surgery,” Shen replies.
Joe thanks him repeatedly.
Shen smiles. “It’s my job,” he says.
Joe looks into Shen’s eyes and thinks: The man is physically exhausted.
He calls Amanda and his parents to give them the news.
Later that morning, Amanda is discharged from Reston Hospital Center. Her sister picks her up and drives her to Inova Fairfax. Amanda remains at Taylor’s side day and night, refusing to leave the hospital until her daughter can.
Three days after surgery, Collazo closes Taylor’s chest. Every day Taylor’s heart grows stronger, and every day Amanda thanks the surgeons as they come by on their rounds.
“They Die in India”
In a softly lit conference room, physicians, nurses, and cardiac technicians gather for a weekly “cath conference” every Wednesday at 5 pm. Here they go over the most troublesome pediatric heart cases—children suffering from devastating congenital heart defects or who have undergone unsuccessful operations at other centers. Dressed in blue scrubs, Shen and Collazo attend along with cardiologists, nurses, and other staff.
“We hold these meetings to go over these difficult cases and try to reach consensus on how to proceed,” says Shen, who chairs them. “We don’t need to discuss the cases where the answers are clear-cut. We also strive to keep our meetings collegial and constructive. They’re not ABC, like at some other institutions.” ABC, he explains, stands for “accuse, blame, and criticize.” Even good hospital cardiac programs can be damaged by finger-pointing in such meetings, Shen says.
For each case, the child’s condition and history of medical interventions is presented by the patient’s cardiologist. All the participating doctors, while affiliated with Inova Fairfax, are in private practice—and in many cases in competition with one another. While this has the potentia
l for turning the meetings into ego clashes, Shen and Collazo make sure it doesn’t.
“The purpose of the cath conference is primarily educational,” Collazo says “because we learn from each other. This is particularly important now that we are taking on many more complex cardiac cases. It’s all about patient outcomes—it’s not for people to get their egos stroked.”
The two dozen or so doctors in attendance see images from three-dimensional echocardiography, CT and MRI scans, and x-ray fluoroscopy on the large screen in the front of the room. To the untrained eye, the images might as well be a Google map of New Jersey, but to trained eyes the grainy images reveal crucial information.
The image of a beating heart appears on the screen. Fine-mesh stents that opened a severely narrowed vessel are visible, as is the rivulet of blood flowing through them. It’s the heart of a young child born with multiple defects who, despite several interventions and numerous medications, still struggles for breath just walking across the street. One cardiologist notes that children with these types of complex heart defects aren’t even operated on in some countries. “They die in India,” he says.
Even though the treatments and medications the child is taking are palliative, the doctors concur that in his weakened condition the risks of additional surgery are too high.
“We’re not talking about getting this child to age 70,” one cardiologist says. “We’re trying to figure out how to get this child to 17.”
They decide that the best course is to wait and see if the child’s condition improves enough to tolerate surgery. “I think we should wait until we have no choice but to intervene,” one doctor says, and there’s general agreement among the others. Perhaps the child’s only hope will be a heart transplant.
Another case flashes on the screen, which shows a dark, circular area in a child’s chest. I ask the cardiologist next to me what it is.
“It’s a pacemaker,” he says. “They used to be the size of hockey pucks, but they are much smaller and much better now, like comparing an old A-frame to a modern computer.”
The physicians discuss other complex cases as well as a death, and as they do I think back to the summer of 1961 at Peter Bent Brigham Hospital, now part of Brigham and Women’s Hospital in Boston, when I came to know two men who were also undergoing heart surgery.
One of them, a genial fellow who expressed little fear, strolled the corridors in his maroon robe, and we often talked in the doorway of his room. We wished each other luck. I still remember the shock I felt when I was told he’d died on the operating table.
Soon after my own operation, I learned that the second man had died a few days after his surgery to repair a defect similar to mine. He was about 35, and I knew that if the technology that saved my life hadn’t been available until I reached 35 or so, I might have suffered the same fate. I suspect that their cases were discussed at Peter Bent Brigham in the same way as the cases being discussed in the conference I witness at Inova Fairfax.
There were no sophisticated cardiac-imaging technologies such as three-dimensional MRIs, CT scans, and echocardiography in 1961—stethoscopes, electrocardiograms, and x-rays were the only diagnostic tools available. Also lacking was the wealth of accumulated experience and knowledge doctors could draw on to better understand the human heart. The survival rate for heart surgery in those days was 60 to 70 percent at the leading institutions.
Today, 95 percent of babies born with congenital heart defects can expect to reach adulthood, and survival rates in pediatric heart surgery at centers such as Inova Fairfax are at or near 100 percent for many defects, making losses today seem that much harder to accept.
>> Next: The Delicate Art of Intervention
The Delicate Art of Intervention
It’s a little after 8 am on a Thursday in September when a neonatal ICU nurse wheels two-day-old Ryder Uhlendorff into catheterization lab 8 in the Heart and Vascular Institute of Inova Fairfax Hospital. Asleep in an incubator and wrapped in a blanket, Ryder barely stirs as the nurse leans over him.
“Bye-bye, baby,” she says softly as she leaves the room.
Russell D’Sa, a cardiovascular invasive specialist and supervisor of the cath lab, lifts the infant onto a table covered with sterile cloth. The lab temperature is 60 degrees, and Ryder cries when he’s exposed to the cool air.
Dr. Robert Mesrobian, section chief of pediatric anesthesiology, leans over Ryder to administer the muscle relaxant propofol through an intravenous line. This allows Mesrobian to slide an endotracheal tube down Ryder’s throat and put him on the ventilator that takes over his breathing. Mesrobian also administers nitrous oxide and sevoflurane, putting Ryder into an anesthetic sleep. The digital monitors above Ryder read out his vital signs—heart rate 169, oxygen level 92 percent. His blood pressure fluctuates but remains in the normal range.
Although dark blue at birth, Ryder is pinker now and looks almost normal. Yet he’s so gravely ill that he would die in days, perhaps hours, without medicine or intervention. Because surgery is inherently risky, Dr. James Thompson, an interventional pediatric cardiologist, will try to correct Ryder’s heart problem with a catheter, but he knows this will be a challenging case.
D’Sa preps Ryder for the catheterization procedure. “You know what they say about interventional cardiology,” he says. “It’s only as good as the jerk on the end of the wire.”
The specific procedure is balloon pulmonary valvuloplasty, developed in the early 1980s at Johns Hopkins by pediatric cardiologist Jean Kan and her associates. Just 37 years old when she devised the procedure, Dr. Kan is now retired and living outside St. Louis. A modest woman who speaks with a soft voice, she seems almost embarrassed to accept credit for her pioneering work when I telephone her to talk.
She says her contribution stood on the shoulders of many, especially the ground-breaking research of Dr. Andreas Grüntzig, a German physician who in the 1970s developed the balloon-catheter technique, commonly called angioplasty, for opening blocked coronary arteries.
Kan says they first needed a larger balloon because the pulmonary artery is bigger than a coronary artery. They then tested the procedure on animals and found that it didn’t set off a fatal cardiac arrhythmia or cause a vessel rupture, their two major concerns. With additional research and permission from the Johns Hopkins investigational committee, Kan and her colleagues performed the first human valvuloplasty in 1981 on Sharon Owens, a young Maryland girl with pulmonic stenosis. The procedure worked perfectly, and her stenosis was cured.
“We followed Sharon for several years, and she never needed to be recatheterized,” Kan says.
The impact of Kan’s research is evident in a recent Pub Med literature search that turned up nearly 3,400 articles on balloon valvuloplasty.
The manufacturer named the device the Owens Pulmonary Valvuloplasty Balloon because Kan didn’t want it named for herself. “I never considered myself a medical pioneer,” says Kan, now in her mid-sixties, “I was part of a continuum.”
While ballooning technology became the first major application of interventional cardiology, over the past 20 years the field has revolutionized the treatment of heart disease in children and adults. Besides pulmonic stenosis, catheter techniques today routinely repair septal defects (the single most common congenital heart anomaly), install vascular stents, open constricted aortas, and are in the beginning stages of replacing malfunctioning heart valves. They’re also used in “hybrid” procedures in ORs in conjunction with surgery to correct certain complex heart defects, and at Children’s Hospital Boston catheter techniques have successfully repaired certain congenital heart defects in utero.
Standing six feet, five inches, well conditioned from running every morning, wearing clogs and a lead apron so heavy that it gives him back trouble from time to time, Dr. Thompson leans in to get a closer look at the monitor displaying fluoroscopic x-ray pictures of Ryder’s beating heart, made clearer with contrast dyes. He shakes his head.
“That’s a very sick-looking right ventricle,” he says. “This is why this case could not be put off for another day.”
In the lead-protected control room just outside the cath room, Thompson points out what he’s talking about.
“See the right ventricle right there?” he says. “See the blood in there, this dark area? It just pools inside the ventricle. It’s not getting pumped out because the pulmonary valve won’t open.”
The first sign that Ryder might have a congenital heart problem appeared in May when his mother, Rebecca, a special-education teacher in the Frederick County public-school system, underwent a routine prenatal ultrasound exam that revealed a single umbilical artery where there are normally two. Though the condition isn’t alarming in itself, Rebecca and her husband, Ryan, learned from the doctors that a single umbilical artery caries a 20-percent risk of a birth abnormality and a 5-percent chance of a cardiac abnormality.
Congenital heart defects are the most common birth defects, affecting 1 in every 125 live births, and even in this era of modern technology they remain the deadliest birth anomaly.
The first ultrasound exam of Ryder’s heart, then about the size of a grape, suggested that he had a relatively mild pulmonic stenosis, or narrowing of the pulmonary artery. This is the same congenital heart defect I was born with in 1939. The pulmonary artery transports unoxygenated blood from the right ventricle to the lungs. If the stenosis is severe enough, the right heart eventually fails from overwork, and death quickly follows.
Although the first glimpse of Ryder’s heart in May suggested a mild defect, pediatric cardiologist James Telep wanted to take a second look when Ryder’s heart was more developed. On July 26, when Ryder’s gestational age was about six months, Dr. Telep studied the ultrasound pictures and observed something not apparent earlier: There was no discernible blood flow through the pulmonary artery. It virtually stopped at the pulmonary valve. Telep knew that the unborn baby had a severe case of pulmonic stenosis.
He told Rebecca that the pulmonary valve had an opening the size of a pinhole, meaning that plans for her baby’s birth had to be changed. He recommended that she have Ryder at Inova Fairfax Hospital because the baby would need immediate medical attention for his heart condition and there was no better place.
Rebecca left Telep’s office trembling. She walked to her car, put her head against the steering wheel, and cried. Ryder wasn’t only her first child but also her parents’ first grandchild.
From her car, Rebecca phoned her mother in upstate New York. In a halting voice, she told her how scared she felt; her mother promised to drive down with Rebecca’s grandmother. Rebecca tried to reach her husband, an electrician, at work, but he didn’t answer his cell phone. At home, Rebec
ca went onto the Internet to learn more about Ryder’s condition.
“I finally had to stop reading about it,” she says, “because I was scaring myself.”
One thing would prevent Ryder from being stillborn. In a developing fetus, a vessel called a ductus arteriosis forms a connection between the pulmonary artery and the aortic arch. This permits unoxygenated blood to bypass the fetus’s fluid-filled lungs and go directly into the aorta to be pumped out to the mother’s placenta to acquire oxygen. The ductus arteriosis normally closes after birth, but researchers found that a naturally occurring hormone called prostaglandin would keep the ductus open when administered to newborns. Prostaglandin was one of the most significant discoveries in pediatric heart care and has saved the lives of countless newborns such as Ryder with congenital heart defects.
“A Light Went Off”
Shortly after birth, doctors give Ryder prostaglandin to allow blood to get to the lungs to be oxygenated. Two days later, Thompson studies the bank of monitors above Ryder’s cath table.
During medical school at the University of North Carolina, Thompson attended a lecture on interventional pediatric cardiology, after which the speaker invited anyone in attendance to cut pathology class to observe the cath-lab clinic. Thompson was the only one to take him up on the offer.
“A light went off that day,” he says, “and I knew I wanted to do interventional cardiology.”
On the monitors, Thompson sees evidence of the damage inflicted on Ryder’s heart during his fetal development. It happened because Ryder’s right ventricle labored with every beat to force blood into his lungs through a pulmonary valve that didn’t open. The fierce resistance causes the ventricle to work ever harder and its walls to thicken, much as any muscle thickens with repeated exertion. With this thickening, called cardiac hypertrophy, the amount of blood the right ventricle can hold is compromised and its ability to beat normally is hindered.
“See how stiff his right ventricle has become?” Thompson says, pointing on the monitor to the ventricle’s sluggish, shallow beats.
On the day I was born in February 1939, in what is now Yale–New Haven Hospital in Connecticut, a young doctor who listened to my chest and heard my heart murmur—a grade five on a scale of one to six—told my mother, “I’m afraid he won’t live beyond adolescence.”
His opinion, cruel as it was to my mother’s ears, reflected the fact that at the time the heart remained off limits to surgical intervention, eliminating any hope of fixing the defect.
I grew up often feeling sorry for myself because I loved sports but wasn’t allowed to play on any of my high-school teams once a doctor listened to my chest.
Now, gazing at the images of Ryder’s tiny heart and seeing how severe his stenosis is and how badly damaged his right ventricle, I understand as I never have before that my pulmonic stenosis could have been much more severe, and if it had been, I might not have lived beyond a few days, much less through adolescence.
Thompson has told Ryder’s parents he’s concerned about sliding a balloon catheter through the pinhole in Ryder’s pulmonary valve to open it up. As he studies the beating of Ryder’s heart on the monitor, his concern deepens. If he can’t open the pulmonary valve with a catheter, Ryder will need surgery.
The room darkens except for the bright lights beaming down on Ryder. X-ray fluoroscopy doesn’t show the heart or other soft tissue very well, but it shows the wire D’Sa has inserted as well as the catheter, so it is necessary. Thompson uses it sparingly to reduce Ryder’s radiation exposure. MRIs don’t emit radiation, but that technology isn’t yet up to this task. Besides radiation exposure, other major risks for Ryder are cardiac arrest, arrhythmia, bleeding, and stroke from a blood clot; in procedures such as this, the unexpected lurks.
On an overhead monitor, a 4 French catheter, the thinnest available for this procedure—and with the even thinner guide wire inside it—can be seen snaking its way up Ryder’s femoral vein on its way to his vena cava, the body’s largest vein, and into the right atrium. From here Thompson, with D’Sa assisting, maneuvers the catheter, which carries a deflated balloon, down through the tricuspid valve and into the right ventricle.
“I hope this pulmonary valve works normally after ballooning,” Thompson says, “but I don’t know.”
The room grows quiet as Thompson tries to position the catheter in the pulmonary valve, a challenge given the small size of Ryder’s heart and the pinhole opening in the valve. If Thompson can work the tip of the wire with a diameter of 14 one-thousandths of an inch through the hole, he’ll be able to position the catheter in the valve and expand the balloon.
When he thinks he has it where he wants it, Thompson inflates the saline-filled balloon.
Nothing.
>> Next: “We Missed It”
“We Missed It”
“The valve is really hard to see, and we missed it,” he says. “I think we were too far in.” He deflates the balloon and tries again. This time, Thompson checks and double-checks the markers on the catheter until he’s satisfied it’s positioned where he wants it.
Thompson inflates the balloon again. On the monitor, the expanded balloon at first appears dark, like two links of sausage amid the gray-and-white opacity of the heart. The narrowing I see between the “links” is where the valve is.
“Watch this,” Thompson says as he pumps more fluid into the balloon. Suddenly, the balloon expands and the narrowing disappears as the balloon rips open the leaflets of the once-shuttered valve. Thompson deflates the balloon quickly.
He stands back and claps his hands. “Yeah, success!” he says, pointing to the blood streaming for the first time through the pulmonary valve on its way to the lungs. I see a dying heart suddenly come alive.
“Balloon coming out,” Thompson says. “How’s the right ventricle doing? I want to see the outflow pressure.”
He watches the pressure drop from 90 to 70, the result he hoped for. The ideal pressure is around 25, and Thompson knows that as Ryder’s right ventricle relaxes in the coming weeks, the pressure across the pulmonary valve will drop into the normal range.
Thompson strides down the hospital’s corridors to the maternity section to talk with Rebecca and Ryan. The couple had Ryder baptized before the catheterization and have spent the morning praying.
“Everything went very well,” Thompson tells them. “As you know, I was pretty worried about what we could do, but I think he’s going to be fine, and I don’t think he’ll need any more procedures, but we’ll have to wait and see on that.”
The couple thanks him profusely. Rebecca, still weak from her C-section two days earlier, calls home to tell her family the results and learns that her Aunt Jane has been saying rosaries and telling God to “keep this baby safe.”
“It must have worked,” Rebecca says.
Ryder remains in intensive care and on prostaglandin for several days while his right ventricle recovers and relaxes after the months of punishment it endured. When he’s eventually weaned off prostaglandin, his right ventricle has to work hard to make up for the closing of the ductus arteriosis. It’s soon up to the task.
After 17 days, Ryder is well enough to be released from the hospital. When I meet with Rebecca and Ryan at their Frederick home a couple of weeks after that, they tell me they didn’t truly feel like parents until Ryder came home. They’ve had a steady stream of family visitors, and people at Rebecca’s school got together to deliver home-cooked dinners every night.
As we talk in their living room, Ryder cries in another room. Ryan goes and picks him up, carries him to the living-room sofa, and offers his young son a bottle. Ryder looks healthy as he takes the bottle and nestles on his father’s chest. He’s slowly being weaned off the beta blockers he’s being given to maintain a steady heart rate.
Thompson has told the couple that another procedure is unlikely to be needed because of the heart’s ability to mend itself. He has told them that Ryder will lead a normal, healthy life.
Like other families I’ve spoken to, they ask about my life after my heart repair, and I assure them I’ve led a normal life in the 50 years since. I married the woman who waited for me during my operation and have had a long and happy marriage, three healthy children, and four healthy grandchildren—the same things they want for their child.
“The Worst Feeling in the World”
Lucas Collazo rises early in his Great Falls home, before his wife, Karen, and their two young children. As he showers, his mind locks in on the surgery he’ll perform that day on a septal defect. He tries to anticipate any surprises.
Collazo is motivated by his perfectionism and his fear of what he calls “the long walk” from the OR to the waiting room to inform a family that a child has died.
“It’s the worst feeling in the world, and it never goes away,” he says. “I can tell you the names and dates of every child under my care that has died. I can’t remember the successes.”
Photographs of little children he’s treated, along with thank-you notes and a Valentine’s card he receives every year from the same little girl, hang on a wall in his office to remind him of his successes. But it’s the losses that drive him.
“I never want to take that long walk again,” he says. “Every time I operate, I know there is a possibility that something unexpected could happen and the patient might die. That thought never leaves my mind.”
After graduating from Columbia University, Collazo, the son of Puerto Rican immigrants, attended SUNY medical school in Buffalo. There, during his first rotation, he watched cardiac surgery and knew he had found what he wanted to do with his life.
The broad-shouldered and barrel-chested Collazo’s hands are so large that, when he graduated from his surgical residency program, his classmates presented him with a bucket of ladyfinger cookies as a joke.
Unlike Shen, who operates exclusively on congenital defects, Collazo also performs adult cardiac surgery for valve replacement, coronary bypass, and other acquired problems. The two men frequently operate together, a practice that’s unusual.
Several independent cardiologists I spoke with over the summer and fall believe Inova has forged one of the strongest pediatric heart programs, if not the strongest, in the Washington area.
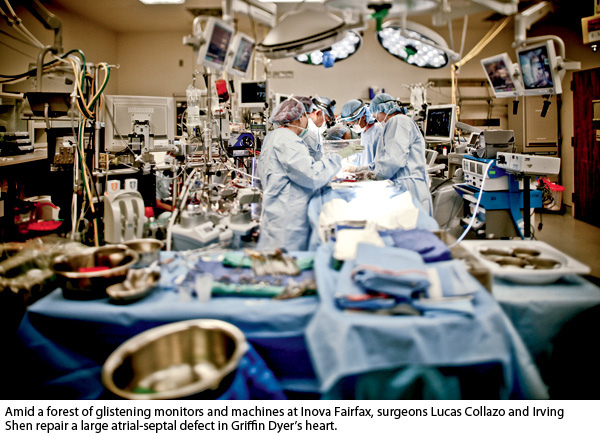
Kari Dyer follows her three-month-old son, Griffin, as he’s wheeled into the operating room while her husband, Cary, remains in the hall outside. A nurse lifts Griffin onto the operating table. Tearful behind her surgical mask, Kari leans down and whispers, “I love you.”
Griffin has Down syndrome, and children with this chromosomal abnormality often have congenital heart defects. Griffin’s hadn’t been detected at the time of his birth on June 7, but his mother became concerned a few weeks later when his appetite waned and his breathing became labored. She took him to pediatric cardiologist Patrick Callahan; an echocardiogram revealed an atrial-septal defect.
Jim Thompson later catheterized Griffin to see if there were any other cardiac abnormalities the scans might have missed. He found none but confirmed that Griffin also had an unusually small mitral valve that would require medical attention. Because of the severity of Griffin’s atrial-septal defect—the infant essentially had no wall separating his left and right atria—Thompson couldn’t repair it in the cath lab.
Because it’s Collazo’s case, Shen stands on Griffin’s left and Collazo on the right. They need to devise a new atrial septum for Griffin. Watching the procedure, I marvel at the speed and ease with which the two talented surgeons operate.
Collazo saws the sternum, cartilage still not yet hardened into bone, down the middle. It makes me uncomfortable: The same thing was done to my sternum 50 years ago, and the painful effects lingered for weeks. I remain grateful for the morphine given me in the days following my surgery.
The surg
eons expose Griffin’s pulsing heart. To see a human heart beating in a chest always seems to convey something sacred. They put Griffin on bypass, cool his blood and body to the low 90s, and stop his heart. Every time I observe this, I think back 50 years to when my heart was stopped while I slept and for a few brief moments I was clinically dead.
Collazo makes an incision in the pericardium, snips out a rectangular patch, and turns it over to physician assistant Thomas Lauer, who dips it into a solution of glueraldehyde. The pericardial tissue is normally floppy, but a few minutes in this solution toughens it the way tanning toughens leather, making it strong enough to patch the hole in Griffin’s septum.
Cutting into the heart muscle, Collazo and Shen expose the area between the atria. Collazo secures the patch cut from the pericardium and attaches it with sutures to what there is of Griffin’s atrial septum. Their fingers move and mesh amid a cat’s cradle of suture threads in the tiny confines of Griffin’s chest.
This surgery is considered “routine” compared with many open-heart procedures, and there are moments of light banter. When the conversation turns to actress Lindsay Lohan, Shen confesses that he has no idea who she is.
“Irving probably has a big poster of her inside his office door,” someone says.
Everyone chuckles, including Shen.
The repair complete, Shen and Collazo watch the pulsing heart in Griffin’s chest as they remove the cannulas and take him off the heart-lung bypass machine. Griffin’s heart starts to beat on its own.
“Sixty-nine minutes on bypass,” the perfusionist notes.
“No surprises on this one, thank God,” Collazo says.
They go through a checklist to account for every instrument, needle, and sponge involved in the operation.
Still asleep from anesthesia, Griffin is wheeled to the pediatric ICU. Collazo meets his parents in a small room. It’s a warm September day, and the couple has walked anxiously around the hospital grounds for more than two hours. Collazo assures them the repair is complete, that Griffin has come through fine, and answers their questions. Cary asks if Griffin will need another septal repair as he grows. Collazo says he won’t, that the tissue surrounding the patch will grow with Griffin.
On an October afternoon three weeks after Griffin’s surgery, I visit the Dyers’ home in Alexandria. Kari speaks softly to Griffin as he nestles in her arms. He’s eating better, she says. Their three-year-old daughter, Lilly, plays with toys on the living-room floor.
Kari says she suffered several miscarriages before Griffin was conceived, so she hadn’t wanted to risk amniocentesis. Everything had appeared normal during the pregnancy, so the Down syndrome came as a shock.
She puts Griffin on the living-room rug. He’s a beautiful baby, round and pink. He takes medication to control pulmonary hypertension. As is often the case in children with multiple heart defects, Griffin’s story is incomplete—he may require additional cardiac intervention someday to repair his mitral valve.
>> Next: 50 Years Later, Two Lucky Men

50 Years Later, Two Lucky Men
Elbert “Bert” Ransom Jr. is impossible to dislike. Gregarious and outgoing, Bert welcomes me to his Alexandria townhouse in August 2010. I’ve come to talk with him at the suggestion of Lucas Collazo, who operated on Bert a few months earlier. Bert and I had similar medical experiences growing up. We quickly developed a kinship based on shared experiences that made it seem as if we’d known one another for years.
Born in Jackson, Mississippi, in 1936, Bert is a longtime civil-rights activist who marched alongside Martin Luther King Jr. during the bus boycott in Montgomery. In 1968, he served as city manager of Resurrection City, the tent city on the Mall that stood as a symbol of the Poor People’s Campaign. Author of two books and an accomplished baritone vocalist, Bert lectures worldwide on nonviolence as an agent of social change under the sponsorship of the US State Department.
In late October 2009, as Bert prepared to travel to Ethiopia on a State Department mission, he routinely strapped his blood-pressure cuff around his arm, pumped it up, and was startled by the digital reading.
“My blood pressure was 211 [mmHg], and I was taking blood-pressure medication,” he says. His systolic pressure should have been in the 120 range. Louise, his wife of 43 years, said, “We’re going to the doctor. Now!”
“If you were my dad,” said the cardiologist who examined Ransom that day, “I’d tell you to cancel that trip and not get on that plane. If something happens to you during the flight, there’s nothing anyone can do for you.”
A few days later, Bert went to Inova Fairfax Hospital for an evaluation of his high blood pressure and what he suspected to be the underlying cause: the reemergence of a congenital heart defect that had been operated on in 1959.
Bert’s family had moved to New Orleans, and at the age of 11 he suffered tonsillitis.
“The doctors at Charity Hospital examined me and detected a heart murmur,” he recalls. “They told my parents they would not operate on me until they figured out the cause of the murmur. I felt perfectly fine at the time. I ran around and played sports like the other kids and never felt any effects from my heart condition.”
Bert told people outside his family about his heart condition, but I never did. I thought my condition was a sign of weakness, so it became my secret.
For years, doctors at Charity Hospital kept reevaluating Bert with chest x-rays and stethoscopes, but nothing unraveled the mystery of his murmur and his high blood pressure. Two months after graduating from Xavier College in New Orleans in May 1959, he moved to Chicago—mostly, he says half seriously, to escape working long hours in his father’s dry-cleaning business. He had a degree in music and applied for a job as a music teacher in the Chicago school system.
“I had to undergo a physical exam to be hired, and the doctor who examined me picked up the murmur. When he did, he looked at me and said, ‘We’re not going to be able to hire you. I suggest you see a heart specialist.’ ”
Bert found his way to Dr. Benjamin Kaplan, who called in other cardiologists and heart surgeon Laurence Rubenstein. In August 1959, Bert was diagnosed with a congenital coarctation of the aorta, a narrowing of the aorta between the aortic arch and the descending aorta. A feature of coarctation is high blood pressure, especially in the upper body.
“I’d never heard the word ‘coarctation’ until that moment,” Burt says.
The doctors recommended that he have surgery to repair the defect because the aortic narrowing put additional stress on the left ventricle to pump blood to the body, and in time the left ventricle would fail.
Bert and I were born with heart defects that seemed to be mirror images of each other. His aortic coarctation overtaxed his left ventricle while my pulmonic stenosis overtaxed my right. So in August 1959, at age 22, Bert underwent surgery to repair his defect. I had my operation almost exactly two years later, in August 1961, also at 22.
Bert’s family didn’t want him to have the “experimental” surgery, but Bert realized, as I did, that the defect had to be corrected if he was to live a normal life, and he was willing to take the risk.
Bert had his surgery at Michael Reese Hospital in Chicago at a time when the heart-lung bypass machine was experimental. “They operated on me without the heart-lung machine by completely icing me and getting my body temperature way down,” Bert tells me.
Collazo explains that when the body is cooled and the metabolism is slowed sufficiently, the aorta can be cross-clamped and the body deprived of blood flow for up to 45 minutes without injury to the brain or other organs. It was the best thing available at the time for many heart patients.
Bert survived the surgery, his aorta was repaired, and his blood pressure dropped into a safe zone. We both have memories of waking from the anesthesia and feeling frozen.
Bert shows me his scar, a horizontal slit in his upper left rib cage. I tell him they split my chest open almost from top to bottom and made a horizontal incision along the top as well, opening me up me like a frog in biology lab. It left a large, T-shaped scar.
It took me years to fully accept that my heart had been fixed. Like Bert, I’d been warned many times not to exert myself, not to run too fast or swim too far because I had a “weak” heart. I remember driving myself to an emergency room two years after my surgery because I felt chest discomfort. All tests came out negative, and I later came to believe that my discomfort had been psychosomatic. It took me four or five years to dispel my demons and stop thinking of myself as fragile.
I suspect most people who have suffered serious medical issues don’t easily escape feelings of vulnerability.
Bert’s heart repair held up for 50 years, until 2009, when his energy seemed diminished and then his blood pressure soared.
With Shen assisting, Collazo operated on Bert the day before Thanksgiving 2009. This time, Bert had the benefit of the heart-lung machine and the skills of Collazo, who navigated through the dense scar tissue in Bert’s chest. When he inserted a Dacron tube and sutured it to the aorta to keep the passage open and maintain a steady flow of blood, Collazo found that the repair made 50 years earlier had scarred over and constricted his aorta again. While he had Bert on the table, Collazo also performed a triple-bypass operation on Bert’s coronary arteries.
As we talk in his living room, Bert tells me that when each of his three children was born, the first question he asked the attending doctor related to the child’s heart. We laugh when I tell him I did the same thing when my three were born. Fortunately, all of our kids came into the world with healthy hearts.
Bert and I share the same lasting gratitude for the doctors who gave us futures. He remains in regular contact with his Chicago cardiologist, Dr. Kaplan, now 89.
I tell him I feel blessed to have had as my surgeon Dr. Dwight Harken, an Iowan who was one of the pioneers of open-heart surgery. He became the first surgeon to operate inside the human heart during World War II when he removed shrapnel from the hearts of wounded GIs in Army field hospitals after the D-Day invasion. In a 1944 letter to his wife, Anne, Harken described the
first time he invaded a beating human heart:
“For a moment, I stood with my clamp on the fragment that was inside the heart, and the heart was not bleeding. Then, suddenly, with a pop as if a champagne cork had been drawn, the fragment jumped out of the ventricle, forced by the pressure within the chamber. . . . [B]lood poured out in a torrent.”
Harken, a lieutenant colonel in the Army at the time, stemmed the torrent and saved the soldier’s life, as he saved the lives of 134 other GIs who suffered potentially lethal wounds to their hearts. He didn’t lose one of them, and all left the hospital with normal, functioning hearts. The first surgeon to repeat his success in a series of cases, Harken shattered the myth that surgeons couldn’t invade the chambers of the human heart, thereby ushering in the era of open-heart surgery that was to spare my life and the lives of many others.
“The Father of Heart Surgery”
I first met Harken, then chief of thoracic surgery at Boston’s Peter Bent Brigham Hospital and a professor of surgery at Harvard, in the summer of 1961, between my junior and senior years of college. I had been referred to him by my cardiologist in New London, Connecticut, near my family’s home in Old Lyme. He thought the time had come for me to be evaluated for surgery.
My cardiologist had been reluctant to refer me earlier because the heart-lung bypass machine, the technological marvel that revolutionized heart surgery, had problems during its early development in the 1950s. The correct temperature for blood circulated through it hadn’t been established, nor had the speed at which blood should be circulated. As researchers tried to figure these things out, the experimental bypass machines triggered blood clots and other complications that caused serious problems for many heart patients, including death. Because I was in good health, I could wait, and by 1961 my cardiologist believed that the pump-related problems had been overcome.
In our first meeting in his office, Harken, a commanding man who exuded both certainty and humanity, instilled confidence in me. He had fiery red hair and, as I came to learn, a temper to match. When he and his young colleagues listened to my heart murmur and studied my heart with fluoroscopy during my first examination, they made educated guesses as to the source of the whooshing noise in my chest, which one doctor said sounded like “a freight train chugging up a hill.” One camp bet on ventricular-septal defect, the other, including Harken, on pulmonic stenosis.
I’ve wondered if listening to my murmur in those days before modern imaging was like listening outside the closed door of a room where people are talking and trying to understand what they’re saying. In any event, a cardiac catheterization—made more worrisome when I learned that a catheter had broken off inside a heart patient the day before and had to be surgically removed—would be definitive.
The rudiments of cardiac catheterization, a technology now in daily use in hospitals in the United States and elsewhere, had been developed in the 1920s by Werner Forssmann, a German researcher who eventually won the Nobel Prize in medicine for this work. But not until the 1950s was the technology safe and reliable enough for patient use. For me, the timing couldn’t have been better.
Dr. Lewis Dexter, an early champion of cardiac catheterization, performed it at Peter Bent Brigham Hospital while I was fully awake and surrounded by men and women in white coats eagerly observing. Dexter inserted the catheter through a vein in my right arm and, using fluoroscopy to guide him, maneuvered it into my right ventricle, where it triggered rapid ventricular contractions every time it touched my heart wall.
I tensed as my heart bounced in my chest like Ping-Pong balls in a lotto drawing. Dexter kept reassuring me that this was normal, but that did little to allay my mounting panic. I’ll always remember a young woman standing near me who squeezed my foot with her hand, a simple gesture that brought me comfort.
The catheter revealed that the pressure of blood flow from my right ventricle through my pulmonary valve and into my pulmonary artery was very high, confirming pulmonic stenosis. The doctors who guessed right couldn’t have been more pleased if they’d picked the winner of the Kentucky Derby.
After the diagnosis, I talked with Harken and told him I regularly played two or three sets of tennis in an afternoon and swam and played pickup basketball games. I think I purposely led a vigorous life in defiance of my fear of the time bomb I knew was beating in my chest. It was my way of asserting my normalcy, of staring down my approaching mortality.
Harken told me that all that activity had resulted in a 20-percent enlargement of my right ventricle. If I didn’t undergo surgery, he said, my health would deteriorate. By my late twenties, I would experience early heart failure, and he didn’t think I would survive much past 35, if that. “You should have the surgery now when you are healthy because it will be far more risky for you once you begin experiencing problems,” he told me.
That convinced me to gamble on possibly losing a few good years if things went wrong versus the chance of gaining a normal life if I survived. My operation was scheduled for August 7, 1961, four weeks after my initial visit to Boston.
Prior to surgery, a number of people in my town volunteered to donate blood both to prime the pump—a bubble oxygenator entangled in tubes that seemed nearly as big as a Volkswagen—and to replace blood I lost. Today blood “lost” in most heart surgeries can be conserved and given back to the patient. The new heart-lung bypass machines, called membrane oxygenators, are the size of small refrigerators. For children, they require less than half a pint of blood for priming. The new pumps have sophisticated software that makes procedures safer for patients. I’m pretty sure the word “software” didn’t exist at the time of my surgery.
I never dwelled on the idea, but thoughts of death intruded when I returned to the hospital. I didn’t fear death so much as I lamented the things I would miss. I loved Lynn, the woman I’m now married to, and wanted us to have a life together.
As the day drew nearer and my tension mounted, Harken strode into my room every morning with his entourage of white-coated residents, all intimidated by him. He once turned to them and asked: “Doesn’t anyone here know how to make a butterfly bandage?” Then he removed one from me and fashioned a new one while they watched in silence.
He would lean down, squeeze my arm with his meaty hand, and announce in his booming voice: “I’m going to save your life.”
The surgery took eight hours. I remember waking up on the operating table to see a gaggle of blurry faces yelling, “Your heart is fixed!” But mostly I remember thinking I was freezing to death.
I remained in the hospital for five weeks, the first few days in a morphine-induced haze. Today, many kids who undergo heart surgery are home in five days.
Harken did what he promised—repaired my heart—and I have had a long and healthy life with, to this point anyway, no cardiac problems. Even my enlarged right ventricle returned to normal size.
In one of life’s coincidences, when I mention Dwight Harken to Irving Shen, he tells me he did his general cardiac s
urgical training under Dr. Alden Harken, Dwight’s son, at the University of Colorado from 1996 to 1999 and considers him his mentor.
“Alden is a brilliant surgeon and scientist,” Shen says.
I saw Harken in his Boston office from time to time in the years following my 1961 surgery. He always told me to consider my heart normal and never let myself become a “cardiac cripple.” I followed his advice. I thanked him publicly in the foreword of a book I wrote and mailed him four years before he died in 1993 at age 83.
Harken’s obituary in the New York Times called him “the father of heart surgery” and also credited him with creating intensive-care units for critically ill patients. The most vivid human being I’ve ever known, he has remained in my thoughts these past 50 years—years I would not have had without his genius and vision.
From my weeks of walking the corridors of Inova Fairfax Hospital and observing its pediatric heart program, I realize that the skill, dedication, and compassion of the physicians and the entire cardiac team offer their young patients the same chance at life Dr. Harken gave me.
Just because repairing heart defects in children is more commonplace today, it is no less a miracle.
This article first appeared in the February 2011 issue of The Washingtonian.